ch6-Categories of Oil Impact
Categories of Oil Impact
Pinnipeds
Most observers of oiled pinnipeds have been unable to correlate the degree of oiling with the type of effect, its consequences, or even associated pathological changes. Nevertheless, we can examine the effects under four broad categories as a background to developing specific action plans. The impacts can be summarized as: a) fur/hair entrapment, b) irritation of eyes, mucous membranes and integument, c) ingestion/ inhalation and systemic effects, and d) behavioral disturbances.
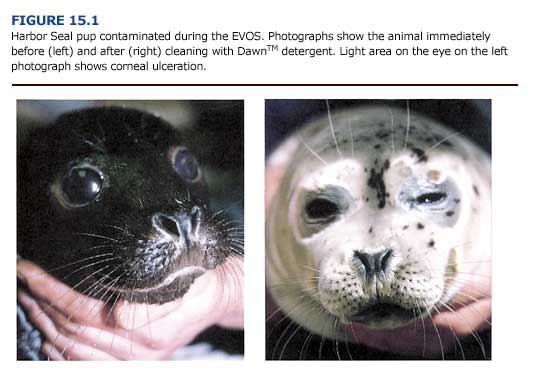
(a) Fur/Hair Entrapment. The direct physical effects of oil entrapped in fur or hair may be severe enough to cause difficulty in swimming, plug nasal passages, and lead to suffocation and drowning. The degree of effect will depend on the degree of oiling, the body surface involved, and the viscosity of the oil (Figure 15.1). Locomotory problems have been observed primarily in young animals such as harp (Warner, 1969) and gray seal pups (Davis and Anderson, 1976) which were so encased that their flippers stuck to the body and the animals drowned. Impaired movements of eyelids and vibrissae may also be damaging (St. Aubin, 1990a). Plugging of respiratory passages with Bunker C oil reportedly killed seal pups at two major spills (Engelhardt, 1985). By contrast, some species, such as the northern elephant seal, can tolerate up to 75% surface fouling with no increase in mortality (Le Boeuf, 1971). In fact, all but one of fifty-eight oil contaminated elephant seal pups were resighted and found to be clean as little as one month later.
Oil fouling of pelage can produce aberrations in thermoregulation. Species that rely primarily on fur rather than blubber for insulation are clearly the most vulnerable. Fur also provides an ideal matrix for oil entrapment. Oil not only subverts the ability of fur to trap an insulating layer of air next to the skin, it also removes the natural oils that contribute to the waterproof quality of the fur (St. Aubin, 1990a).
Thermal demands are greatest for polar and subpolar pinnipeds. However, these species depend primarily on blubber for insulation with the result that external contamination causes little thermoregulatory stress. This distinction applies to adult animals only, as most neonatal pinnipeds initially rely on lanugo for insulation until they develop an effective blubber layer. The degree to which the pelt is compromised might also differ in molting animals, which experience some degree of physiological stress at that time.
Studies that measured changes in heat conductance through pelts have shown up to a 50% decrease in the insulating value of fur seal pelts oiled in vitro. A smaller decrease was found in oiled pelts from Weddell seal pups, and virtually no change was reported for the relatively poor insulating pelts of sea lions, bearded seals, and ringed seals (0ritsland 1975; Kooyman et aI., 1976, 1977). These findings do not address adaptive responses, such as shivering, alterations in metabolic rate and changes in peripheral circulation that may alter the thermal balance of living animals.
(b) Eyes, Mucous Membranes, and Integument. Oil is an irritant and can produce acute changes to the mucous membranes of the eye, oral cavity, respiratory surfaces, and anal and urogenital openings. The extent of damage will depend on the duration of exposure and the volatility of the oil. Most reports of oil-fouled pinnipeds include descriptions of excessive lacrimation, acute conjunctivitis, swollen nictitating membranes, corneal opacity (Figure 15.1), abrasion, and occasionally ulceration (e.g. Lillie, 1954; Spraker et aI., 1994; Smith and Geraci, 1975). The latter authors noted that most signs subsided when the animals had access to clean water.
The integument is less sensitive to contact with petroleum hydrocarbons, and pathological changes have rarely been reported in this organ. This may be due to the limited amount of available data. However, it is clear that animals with sparse pelage, and perhaps those during the molt, would be most vulnerable. Johnson (1983) noted that the creviced skin of the walrus may present special problems. The degree to which hydrocarbons can be absorbed across the integument is unknown, but Engelhardt (1985) felt it was unlikely to make a longterm contribution to tissue burdens.
(c) Ingestion, Inhalation and Systemic Effects. Ingestion of petroleum hydrocarbons adhering to the body surface is of little concern to pinnipeds, which groom using their flippers rather than their mouth. Thus, pinnipeds are only likely to ingest oil by: 1) accidentally opening their mouths while swimming, 2) sucking from an oil fouled nipple or playing with contaminated debris, and 3) ingesting contaminated prey. The first two routes are unlikely to lead to significant intake, and the third only applies to pinnipeds such as walruses and bearded seals, which consume benthic invertebrates, prey items that are known to accumulate hydrocarbons (McLaren, 1990).
Three limited studies demonstrated that phocid seals fed small quantities of oil were not obviously affected, even though the oil was readily absorbed through the intestine and distributed to body organs (Smith and Geraci, 1975; Geraci and Smith, 1976; Engelhardt, 1982). Conversely, oil in the gut and other organs was considered the cause of death in some gray and harbor seals stranded in France (Babin and Duguy, 1985). A causative relationship was not established in the latter study because of the autolyzed state of the carcasses. Regardless, most data indicate limited ingestion of oil by pinnipeds and an ambiguous clinical expression of metabolic toxicity from the ingested oil.
Hydrocarbons can also be absorbed by inhaling vapors. Experiments have shown that pinnipeds can accumulate petroleum hydrocarbons up to several ppm in blood and tissue after exposure to petroleum vapors (Engelhardt et aI., 1977). Following the oil spill from the Sanko Harvest, volatile compounds (particularly acetone) in the blood reached levels of between 30-90 ppm in fur seals that encountered the oil within twenty-four hours of the spill. This compares with 10-20 ppm in those animals exposed to the oil after it had weathered for more than seventy-two hours. Presumably, most of the volatile compounds had dissipated by this time. The concentrations of straight chain petroleum hydrocarbons in these same animals were less than 1 ppm (Gales, 1991). Based on these studies, it appears that inhalation or perhaps dermal absorption, rather than ingestion, were the primary routes of exposure in the fur seals. Once again it was not possible to establish a causal relationship between the degree of exposure (as determined from petroleum hydrocarbons levels in the blood) and mortality or tissue pathology (Gales, 1991).
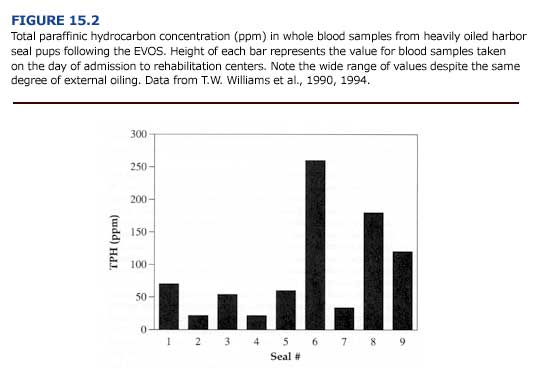
Likewise, harbor seal pups exhibited few pathologic injuries and low mortality despite heavy external oiling following the EVOS (T. M. Williams et aI., 1990, 1994). As found for sea otters, total paraffinic hydrocarbon concentrations in the blood were variable for the pups (Figure 15.2). Values ranged from 22 ppm to 260 ppm (mean = 91%B127 SE ppm, n = 9). Although some of the seal pups had blood hydrocarbon levels above the calculated lethal threshold dose for sea otters (Chapter 4), none showed evidence of systemic toxicosis and all survived. The comparatively high survivorship of the seal pups is undoubtedly related to many factors including: 1) age, 2) species-specific differences in the uptake, metabolism, excretion, and storage of petroleum hydrocarbons, and 3) oil weathering prior to contact (T. M. Williams et aI., 1990).
It is likely that pinnipeds can detoxify absorbed hydrocarbons in a manner similar to other mammals, but may suffer damage to various organs if blood or tissue levels reach critical thresholds (St. Aubin, 1990a). We will discuss pathological and clinical findings in a later section and refer readers to Neff (1990), St. Aubin (1990a), Frost et al. (1994) and Engelhardt (1985) for general details of petroleum hydrocarbon toxicity and clearance in pinnipeds.
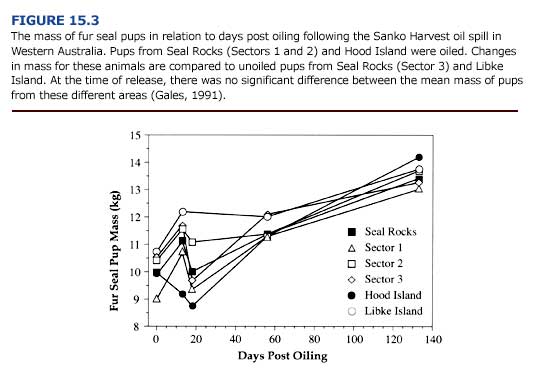
(d) Behavior. Oil spills can disrupt normal behavior by restricting movements to and from haulout sites. We do not know whether pinnipeds can detect oil, and if they can, whether or not they will avoid it. Although there is no experimental evidence that fouling with oil affects behavior in pinnipeds, there have been several anecdotal observations that infer some disturbances. Uncharacteristic swimming behavior, including vigorous head shaking, and swimming with the neck, head and trunk out of the water, has been noted in fur seals occupying a chronically polluted harbor (Shaughnessy and Chapman, 1984). Further, there is contradictory evidence that the mother-pup bond, for which scent appears to be an important component, may be disrupted. Observations of pup rejection by females has been noted, although the familial relationship between the observed animals was not known. Davis and Anderson (1976) reported that the mother-pup bond in gray seals was not affected by oiling, and noted that interrupting feeding to clean oiled pups may have had a greater impact on eventual mass at weaning than did the oiling itself. Heavily oiled New Zealand fur seal pups were observed nursing from females; there was no significant difference in the preweaning mass of heavily oiled pups and unoiled pups (Figure 15.3; Gales, 1991).
The behavior of different age classes of pinnipeds will determine their chances of encountering oil. For example, fur seal pups often congregate in tidal pools where there is little wave activity and oil can become trapped. The risk of fouling is greater for pups than for adults. Older animals tend to enter and leave the water in areas of higher wave activity where oil is physically repelled from the rocks. This basic difference in behavior explains why, after the sinking of the Sanko Harvest, an entire cohort of pups was contaminated, but no adults came into contact with the oil (Gales, 1991). There is clearly a need for quantitative studies to clarify the behavioral impact of oil fouling.
Polar Bears
A single laboratory study (0ritsland et aI., 1981) highlighted the vulnerability of polar bears to oil. Like sea otters and fur seals, polar bears depend on fur for insulation. Fouling of the coat increases heat loss, placing the animals in thermal stress. Polar bears also share the sea otter’s compulsion to maintain a clean coat, and will ingest any oil adhering to their fur. In two of three captive bears, the combined effects of metabolic stress and hydrocarbon toxicity had fatal consequences. Liver and kidney failure, anemia, and depressed lymphoid activity were noted. Secondary bacterial and fungal infections, which contributed to the animals’ demise, were evidence of stress-related impairment of immune function. It is unknown whether bears exposed in the wild would exhibit the same complications in the absence of the additional stress of captivity.