ch8-pregnancy
Pregnancy in Sea Otters
Few reports provide comprehensive data concerning the rate of success for delivery and rearing of pups by wild sea otters. It has been estimated that only 30% of pups survive their first year (Jameson and Johnson, 1993; Reidman and Estes, 1990). Immature females are considered less capable than experienced adults in meeting the constant demands imposed by feeding and grooming of newborn pups.
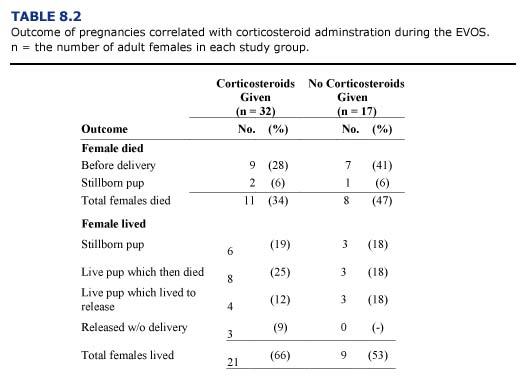
During the wildlife rehabilitation program following the EVOS, nearly 70% of the captured sea otters were female; forty-nine of these were diagnosed as pregnant during admission or at necropsy. In forty (81%) of these females, pregnancy was terminated by: 1) death of the mother (n = 19); 2) abortion of a near term fetus or stillbirth (n = 9); or 3) death of the newborn pup (n = 12). Thirteen newborn pups were transferred to a nursery after their mothers were unable to care for them; all but one of these pups died. Four pregnant females completed the rehabilitation process and were released before delivery. Only five of the pregnant otters were able to deliver and successfully care for live pups in the rehabilitation center (Tuomi et al., 1991).
Mortality in pregnant otters reached 38% and was 4% higher than the overall mortality of captured otters following the EVOS. Pregnant otters admitted during the first three weeks exhibited the greatest degree of oiling and suffered the highest mortality. This was similar to results reported for all sea otters in the rehabilitation program (Williams et al., 1990). Hepatic and renal lipidosis was 2.6 times more common in females than males. Females may have been predisposed to these disorders by the high energy demands of pregnancy and lactation (Lipscomb et al., 1993).
Stillborn pups ranged from 1.2-2.0 kg in body weight; five of these pups showed lesions suggesting death in utero before the onset of labor. Neonatal death (pup survival less than three days) occurred in ten of the liveborn pups; eight of these pups weighed less than 1.4 kg and may have been born prematurely. Samples of tissue from a deceased pup and colostrum from one of the females revealed significant levels of petroleum hydrocarbons (see below). One pup had a large umbilical defect with evisceration and died within a few minutes. Another pup underwent successful surgical repair of an umbilical hernia at two days of age. Uterine torsion was diagnosed postmortem in three females with very large full-term fetuses (fetal weight = 2.0, 2.3, and 2.5 kg).
Nine females brought to rehabilitation centers were accompanied by live pups ranging in age from a few days to several months. Two of the nursing females died and their pups were moved to a nursery along with three more pups from this group whose mothers could not care for them. Two of these five separated pups died.
Eleven other orphaned pups were captured and brought into the centers. Orphan pups from the wild had a lower mortality than pups born in the rehabilitation centers. Ten (91%) of the original eleven wild orphaned pups survived and were transferred to seaquaria.
In comparison to the sea otters from the EVOS, reports from a fifteen-year captive breeding program at the Vancouver Aquarium indicate a 72% success rate with eight pups raised to weaning. One stillbirth, one neonatal death, and one pup lost due to lack of maternal care occurred in this series.