Cages, Pens, and Pools
Sea otters may be housed in portable cages, pens, pools, or floating pens, depending on their health and the condition of their fur. The rehabilitation process can be divided into three phases, each with its particular type of housing: critical care, recuperation, and rehabilitated and awaiting release.
Critical Care
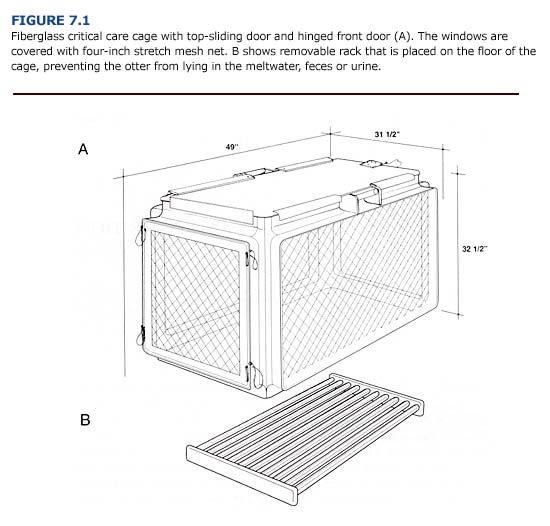
Portable cages with top and side-mounted, sliding doors (Figure 7.1) should be used to hold sea otters during triage, while they recover from sedation, and when they are seriously ill and require frequent veterinary care. These cages are normally used in the triage room and critical care room of the rehabilitation facility (see Chapter 12) and may be used to transport sea otters over long distances. The cages should be made of smooth fiberglass and have a removable rack that allows the passage of water, feces, and urine into the cage bottom. The interior surfaces of the cages should be rounded to prevent the sea otters from damaging the cage or breaking their canine teeth. The windows should be covered with four-inch, stretch mesh net (0.12 inch braided nylon cord). Each cage should be equipped with handles at the corners (placed away from the windows) to allow the staff to safely carry the caged otter to various areas in the facility.
The critical care room should be well ventilated to reduce humidity and odors, and climate controlled to maintain a temperature of 15°C (60°F). When weather permits, sea otters recovering in critical care cages can be placed outdoors with appropriate provisions for shade and windbreaks.
Recuperation
Sea otters should be moved to pens with seawater pools (Figures 7.2 and 7.3) as soon as they are eating, can maintain a stable core temperature, and have no other serious clinical disorders. These pens, which can hold two adult sea otters, are made of fiberglass and have four doors so that husbandry personnel can quickly net an animal when clinical care or relocation is required. As with the critical care cages, the interior surfaces should be rounded to prevent broken teeth. The large windows and doors, which are covered with four-inch stretch mesh net (0.12 inch braided nylon cord), provide good ventilation. The pliable net also prevents the otters from damaging their teeth and gums if they bite it.
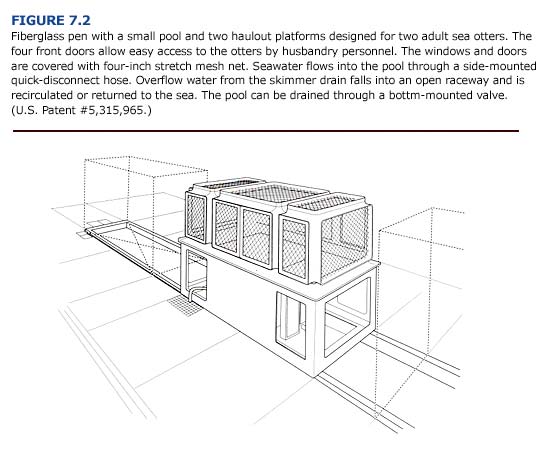
The pens should be located outdoors in an area with low visual and acoustic disturbance. In Alaska, or where air temperatures drop below freezing, pens may be placed in a climate-controlled indoor area and used for otters that are unable to maintain a normal stable core body temperature.
The seawater pool in each pen should be large enough (3 feet square and 2 feet deep) to allow the otters to perform normal grooming behaviors such as rolling and tumbling. This behavior is essential for the restoration of the fur (see Chapter 6). Because otters defecate frequently, the seawater flow into the pools should result in one complete turn- over every thirty minutes (i.e. about 5 gallons per minute in a 150 gallon pool) to prevent fouling the fur and high concentrations of coliform bacteria. The pool should have surface-skimming drains to eliminate floating debris. Clean seawater should be used in the pens and pools to promote the optimum recovery of the fur. If the seawater supply to the pens and pools is recirculated, it must be filtered and sterilized by chlorination or ozonation (see Chapter 12). Because chlorine will damage the otter’s fur, the water must be dechlorinated before it is returned to the pens and pools. Non-chlorinated fresh water can be used for several days if seawater is unavailable, but the long-term effects of holding sea otters in fresh water are unknown.
The pool seawater temperature should be similar to ocean temperatures representative of the season and the sea otter’s geographical home range. However, severely debilitated otters that have lost the thermal insulation of their fur will chill rapidly in cold water. Warming the pool water to 20°C (68°F) with a heat exchanger may allow otters with damaged fur to groom for longer periods before they begin to chill. In theory, this could decrease the rehabilitation time. As the otters restore the water repellency of their fur, the water temperature should be lowered gradually to the normal ocean temperature for that region. Most facilities are unable to regulate seawater temperature, so the potential benefits of warming seawater in the pens remain untested.
The haulout platforms in the pen should be made of fiberglass that is smooth and has beveled holes (0.4 inches in diameter spaced every 3 inches) for water drainage. The haulout area should be large enough (1.7 feet wide by 3.5 feet long) for an adult otter to move about comfortably and continue grooming after emerging from the water. A removable lid of smooth fiberglass should be placed over the pool to prevent the otter from reentering the water if it becomes chilled or when the air temperature is excessively cold.
Recuperating otters may spend much of their time on the haulout platforms and are prone to abrasions and decubital sores, especially on the flippers and rear legs. A smooth, perforated surface lessens the incidence of these lesions. Materials such as wire mesh, plywood, rigid plastic grate, and indoor-outdoor carpeting are unsatisfactory. Once sores appear, they usually persist until the otters are able to spend the majority of their time in the water.
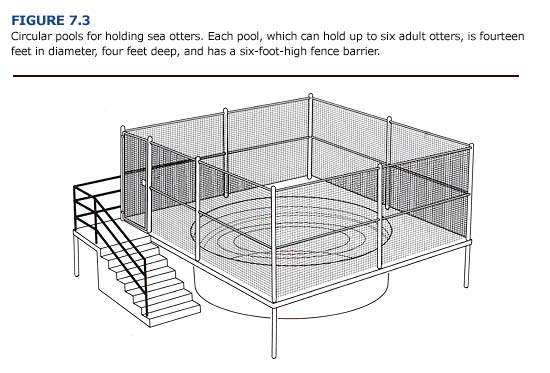
Otters that are eating well, show normal grooming behavior, are able to thermoregulate in seawater at ambient temperatures, and have no other health problems should be moved to larger outdoor pools. The larger area allows the otters to swim, dive, groom, and socialize more actively than in the smaller pen pools. These outdoor pools are circular (14 feet in diameter and 4 feet deep) and can hold up to six adult otters. Smooth haulout areas (3 feet wide) should be provided around the perimeter of the pools. The design should allow the husbandry staff to net an otter easily if clinical treatment is required. The seawater supply should result in one complete turnover every thirty minutes to prevent high concentrations of food, fecal debris, or coliform bacteria.
Sea otters that have been cleaned of oil may successfully restore the water repellency of their fur in one to two weeks (see Chapter 6). Normal grooming behavior is an essential part of the rehabilitation process. Not surprisingly, otters that have been heavily oiled or have other serious health problems may not groom effectively and will require additional time to restore their fur. Improper sanitation or inadequate seawater turnover also will delay recovery.
Rehabilitated and Awaiting Release
As soon as oiled sea otters have regained their health and the water repellency of their fur, they should be moved to a prerelease facility. The prerelease facility consists of large, floating pens (see Chapter 12) located in a clean bay or lagoon with good seawater circulation. Each floating pen should be sufficiently large (at least 18 feet long, 10 feet wide and 5 feet deep) for the otters to actively swim and dive to regain their stamina, muscle tone, and respiratory capacity. On average, each otter should have at least thirty square feet of surface area in the floating pen and six square feet of haulout space.
Animal Care
The otters should be monitored twenty-four hours a day by qualified personnel who are familiar with normal sea otter behavior and can recognize clinical signs of distress. Monitors should be assigned to specific animals or pens (one to four otters in critical care or up to ten animals in the pools and pens). They are responsible for feeding, record keeping (Appendix 2, Forms J-Q Download PDF), maintaining cleanliness, and ensuring that the otters are able to groom effectively. Abnormal behaviors should be noted and corrective action taken immediately.
Animals should be handled by trained personnel and only when necessary for transport, treatments, or sanitation. The physical and chemical restraint of sea otters is described in Chapter 3. During routine husbandry procedures that require moving an otter from one cage or pen to another, the staff should use a large dip net fitted with a soft mesh net (4.5-inch stretch mesh). Otters should be weighed during these transfers by suspending the net from the hook of a hanging spring scale. Alternatively, the otter can be placed in a cage and weighed on a platform scale. An otter’s weight may vary by several pounds, depending on whether the coat is wet or dry. Weighing an otter only when dry or noting a “wet weight” will avoid confusion in the animal’s record.
The husbandry staff should avoid loud talking or socializing near the pens and should observe the otters from a distance or behind a blind to reduce stress on the animals. At the same time, adequate access to the pens is needed for monitors to feed and assist debilitated animals.
It is imperative that the fur remain clean and free from contamination by food or excrement. If an otter in the critical care cages becomes soiled, it should be rinsed in the cage with fresh water or seawater from a low pressure hose. Haulout platforms also should be rinsed of food or feces, and all nets, gloves, and transport cages thoroughly cleaned between use.
Hypothermia is a potentially fatal complication when otters are held out of water (see Chapter 5). In the critical care cages, as soon as otters are dry and no longer at risk of hypothermia, about one-third of the cage floor should be covered with several inches of clean, chipped ice to prevent overheating. Ice helps to prevent pressure sores and keeps the otter’s fur clean. Sea otters like to chew on ice, and this behavior also may alleviate stress and dehydration. Cool water sprays may be used to prevent overheating and shade should be provided when necessary in outdoor areas.
Otters that become chilled in seawater pools or pens, have difficulty hauling out, or experience seizures should be removed from the water with dip nets. If an otter appears hypothermic (begins to shiver violently or becomes lethargic) it should be placed in a critical care cage or confined to a haulout, dried with room temperature air from pet dryer, offered food, monitored for signs of abnormal behavior, and evaluated by a veterinarian.
Rehabilitated otters awaiting release require only a small husbandry staff (one person for ten otters) to feed and monitor them, maintain sanitation, and provide for security. Under normal circumstances, prerelease otters should be handled only when introducing or removing them from a pen, or for occasional clinical procedures.
Toys
Grooming behaviors will occupy most of the time spent by sea otters in the rehabilitation facility. However, otters near recovery appear to benefit from access to objects they can chew and manipulate. Blocks of ice, clam shells, kelp, or artificial toys may all serve to occupy them and satisfy their need to manipulate objects in their environment. Artificial objects (pet chew-toys or net buoys) should be large and tough enough to avoid accidental ingestion.
Social Grouping
When placing sea otters in pens or pools, consideration must be given to social groupings. Otters are naturally gregarious animals that frequently gather into large rafts of up to several hundred individuals in favorable feeding or resting locations. Grouping otters within a rehabilitation facility appears to increase grooming activity and appetite, and reduce signs of stress. Even visual contact between sea otters can be beneficial, but housing two or more compatible animals in a pool or pen is preferred.
Adult males and females should be separated to prevent injury to females during copulation. Juveniles of both sexes can be held with mature females. Placement with these older animals often results in bonding, which mimics natural social patterns and may help younger animals learn normal behaviors prior to release. Dominant males may fight if two or more are placed in the same pen or pool with females; however, groups of up to twenty males have been held in large floating pens without problem (Tuomi, 1990). Pregnant females and females with pups need to be housed in smaller groups and observed more closely (see Chapter 8). Once formed, social bonding appears to contribute to the health and well-being of otters in the rehabilitation facility. Movement of otters out of stable groups should be avoided until final release.
Transport
During short duration (less than six hours) transport outside of the rehabilitation facility, otters should be placed in kennel cages equipped with a durable, tight fitting rack made of PVC pipe (1.5-inch-diameter spaced one inch apart) in the bottom. The rack should be at least two inches above the floor of the cage to allow water, urine, and feces to fall away from the otter. Chipped or block ice should be placed in the cage to prevent overheating and as a source of fresh water. For long trips (six to forty-eight hours), the otters should be transported in the critical care cages (Figure 7.1). The top-mounted sliding doors allow easy access to the animals by a veterinarian or animal care specialist. Adequate food and ice should be carried in an ice cooler during transport so that the otters can be fed every three hours. Spray bottles of water should be used to flush food, feces, or urine from the fur as needed and to prevent overheating. The air temperature in the vehicle or aircraft should be maintained below 15°C (60°F) (Williams, 1990).





